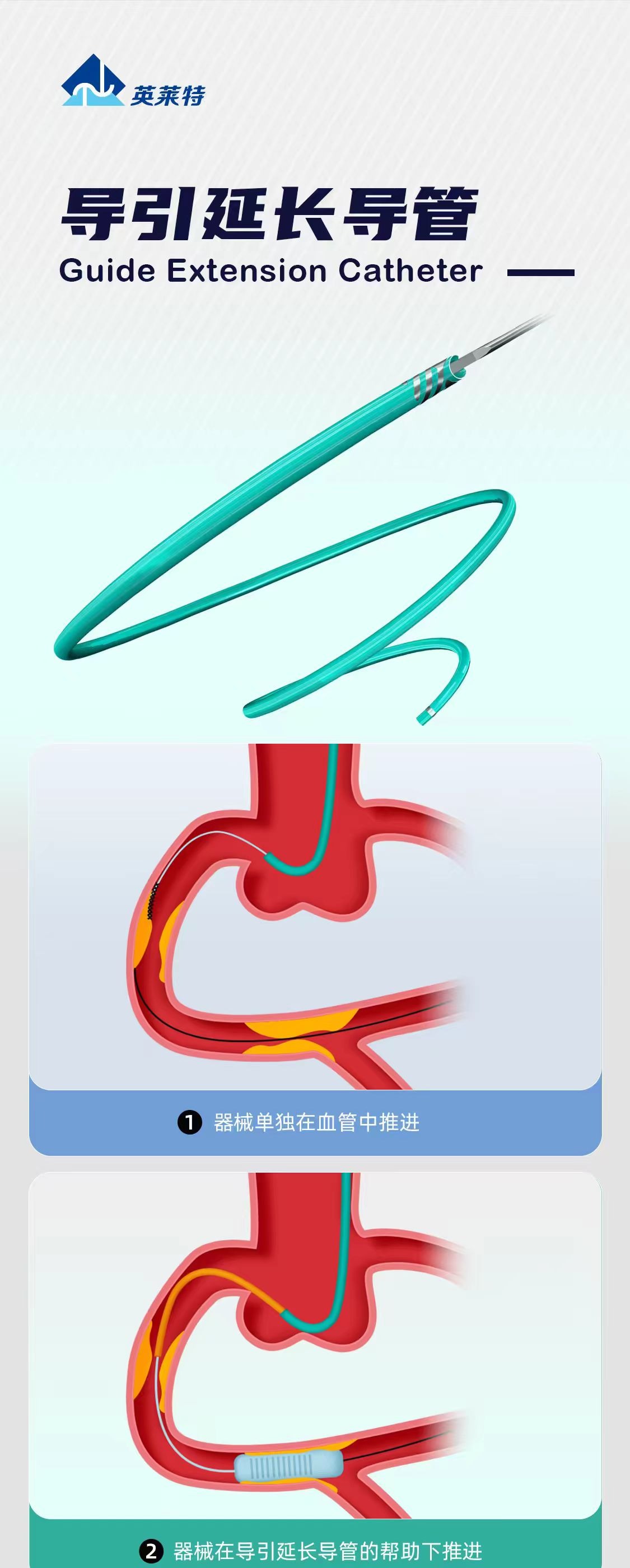
Guide extension catheters are often reduced online to a simple phrase such as “access support catheter.” In reality, their role is much more specific. Based on public Guidezilla II references, complex coronary intervention literature, and your current brochure assets, the real value of this product class is not “extension” by itself, but improved support and more reliable delivery of balloons, stents, and other devices when standard guiding support is not enough.
1. Why complex coronary lesions need this kind of device
In complex PCI, device-delivery failure is rarely caused by one factor alone. It often reflects a combination of:
- heavy calcification
- tortuous vessel course
- distal lesion location
- poor take-off angle or anomalous origin
- limited support in radial access
- guide catheter back-out during advancement
That is where a guide extension catheter becomes valuable. By advancing further into the vessel pathway, it shortens the unsupported route for the interventional device, improves back-up support, and helps balloons or stents reach the lesion more smoothly.
This same logic appears consistently across public material for Guidezilla II, GuideLiner, and similar devices: the product is not meant to replace the guiding catheter, but to strengthen device delivery in complex anatomy.
2. The real procedural logic is “more support, shorter delivery path, better deliverability”
If the website wants to explain the product well, it should not stop at saying “adds support.” The stronger story is procedural:
- the guiding catheter establishes base access
- when complex anatomy makes standard support insufficient, the guide extension catheter is advanced
- deeper support shortens the unsupported segment for balloons, stents, and other devices
- the result is better deliverability and smoother procedural continuity
That is why the website should lead with the use scenario, not the parameter table.
3. Coronary intervention is the main scene, but the distal-delivery logic also appears in peripheral use
Based on Guidezilla II style references, the primary scene remains complex coronary intervention, especially:
- complex lesions
- calcified lesions
- tortuous vessels
- distal lesions
- radial-access procedures needing stronger support
- anomalous coronary origins or unfavorable anatomy
At the same time, public literature shows that the same guide-extension logic has also been used in selected peripheral interventions, including infrapopliteal lesions. In those reports, similar devices were used to improve distal access, sustain pathway support, and help device delivery in difficult below-knee anatomy.
The right website wording, therefore, is careful but useful:
- complex coronary intervention is the clearest and most established scenario
- distal device-delivery support is the deeper logic of the product class
- below-knee or distal peripheral mention should be framed as a literature-supported use logic for similar devices, not as an expanded formal indication unless your registration materials confirm it
4. Specifications and compatibility determine whether the product is actionable
Guide extension catheters are a classic example of a product class that looks simple but depends heavily on fit and pairing. Once a user understands the clinical value, the next questions are practical:
- model
- working length
- compatibility platform
- deliverable device types
- best-fit anatomy
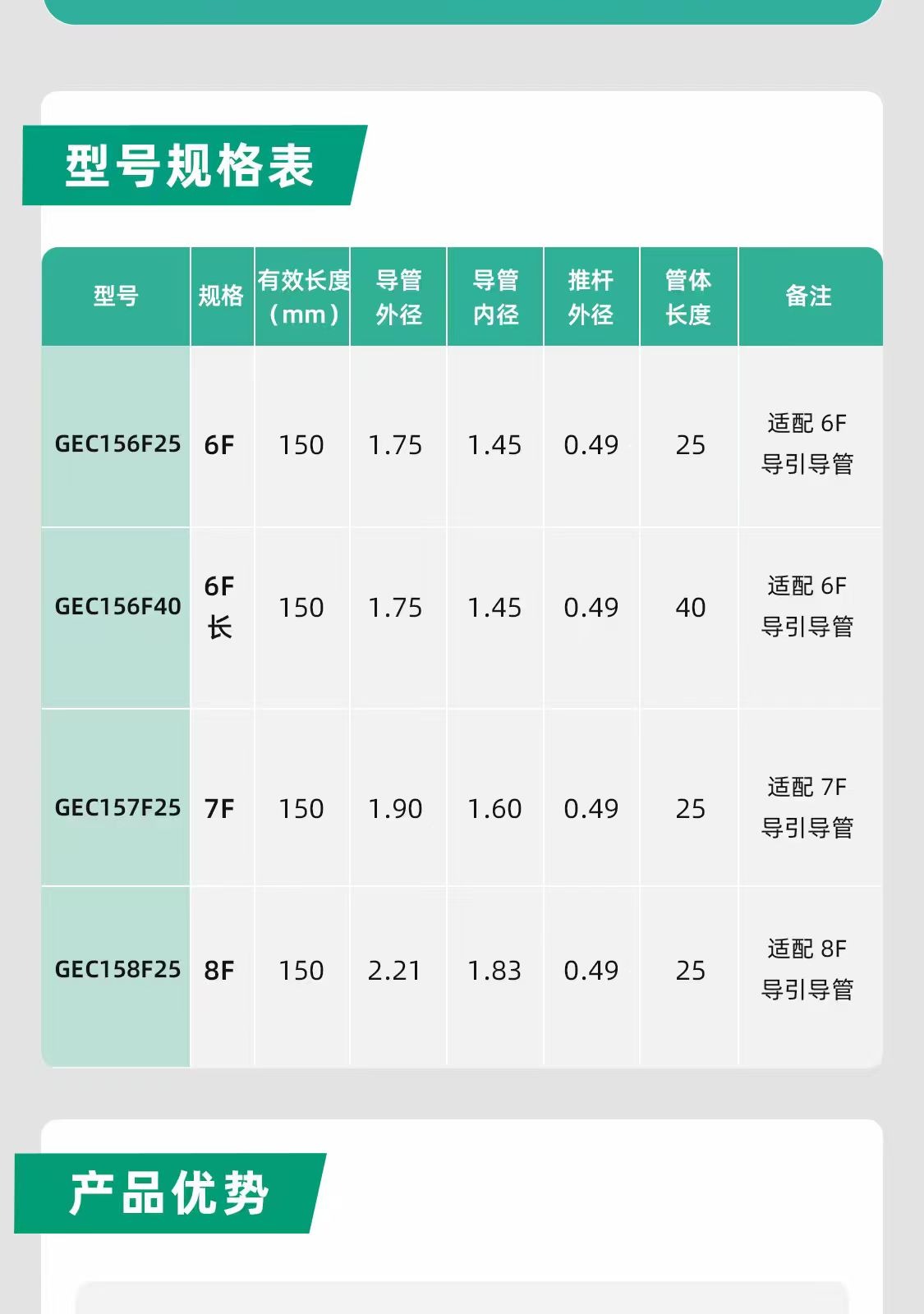

For that reason, the website should always separate the clinical logic block from the specifications and compatibility block. The first explains why the device is used. The second explains whether it fits the case and platform.
5. A better article structure for the website
The most useful website framework for a guide extension catheter usually answers these questions:
What problem does it solve
Standard guide support is insufficient in complex or distal lesion pathways.
Why does it become necessary during the procedure
Because deeper support and a shorter unsupported device path improve deliverability.
Which scenarios are most typical
Complex PCI, calcification, tortuosity, distal lesions, radial access, anomalous take-offs, and literature-described distal peripheral delivery scenarios.
What will the user want next
Specifications, compatibility, supported device types, and a formal materials-request path.
6. Why this matters for the wider website
When the guide extension catheter is explained well, it becomes more than a product-list item. It becomes:
- a representative access-support product for complex coronary intervention
- evidence of the company’s understanding of catheter-based intervention workflows
- a useful anchor for more educational and search-oriented content
Future website topics can build around:
- support strategy in complex PCI
- why calcified and tortuous lesions challenge device delivery
- how radial access changes backup-support needs
- distal lesion and below-knee device-delivery logic
Literature and References
These references support industry-level interpretation. Final indications and use boundaries should follow formal registration and IFU documentation.
- Boston Scientific. Guidezilla II Guide Extension Catheter.
https://www.bostonscientific.com/en-US/products/catheters--guide/guidezilla-II-guide-extension-catheter.html - Boston Scientific. Guidezilla II IFU.
https://www.bostonscientific.com/content/dam/elabeling/ic/guidezilla_ii/51395899-01A_GUIDEZILLA_II_IFU_ML_s.pdf - Guide extension catheters: Review of technology and future directions.
https://pmc.ncbi.nlm.nih.gov/articles/PMC10718559/ - Usefulness and Safety of a Guide Catheter Extension System for the Percutaneous Treatment of Complex Coronary Lesions by a Transradial Approach.
https://pmc.ncbi.nlm.nih.gov/articles/PMC5588208/ - Usefulness of a Guide Extension Catheter in Endovascular Therapy of Infrapopliteal Lesions.
https://pubmed.ncbi.nlm.nih.gov/32018022/